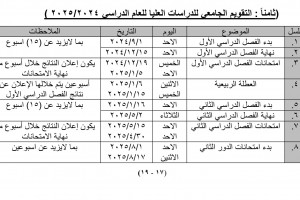
Preparation and Characterization of the Biological Activity of Some Isoxazolidine Derivatives
The College of Pharmacy at the University of Basrah successfully held the defense of a Master's thesis entitled: "Preparation and Diagnosis of the Biological Activity of Some Isoxazolidine Derivatives", submitted by researcher Wijdan Abbas Naima.
The thesis involved the preparation of 1,3-dipolar cycloaddition of nitrone with N-phenylmaleimide to synthesize 8 isoxazolidine derivatives. These compounds were characterized using advanced spectroscopic techniques including:
- 01 Infrared Spectroscopy (IR)
- 02 Proton Nuclear Magnetic Resonance (1H NMR)
- 03 Carbon-13 Nuclear Magnetic Resonance (13C NMR)
- 04 Mass Spectrometry (MS)
The 1H NMR spectra of the isoxazolidines revealed a doublet signal in the range (4.94-5.48) ppm attributed to proton H-6a with a coupling constant of 7.33-9.23 Hz. The 13C NMR spectra showed 3 new chemical signals in the aliphatic region confirming the cycloaddition between maleimide and nitrone.
The study aimed to evaluate the prepared compounds in vitro for their anticancer activity against the human MCF-7 breast cancer cell line using the MTT assay.
.jpeg)

Postgraduate Studies Committee
College of Pharmacy | University of Basrah